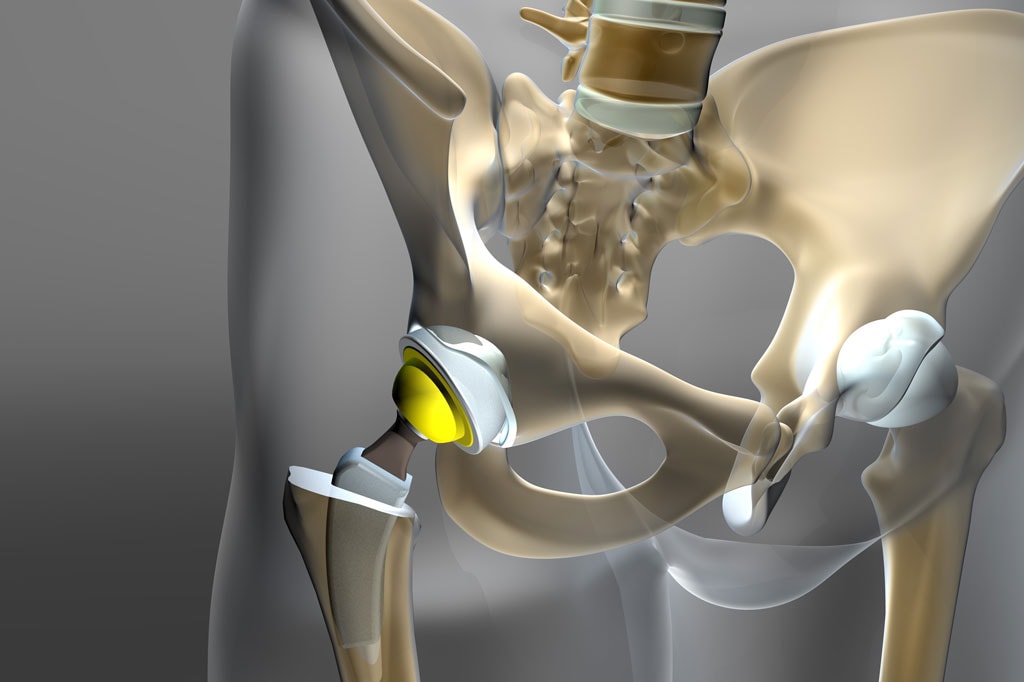
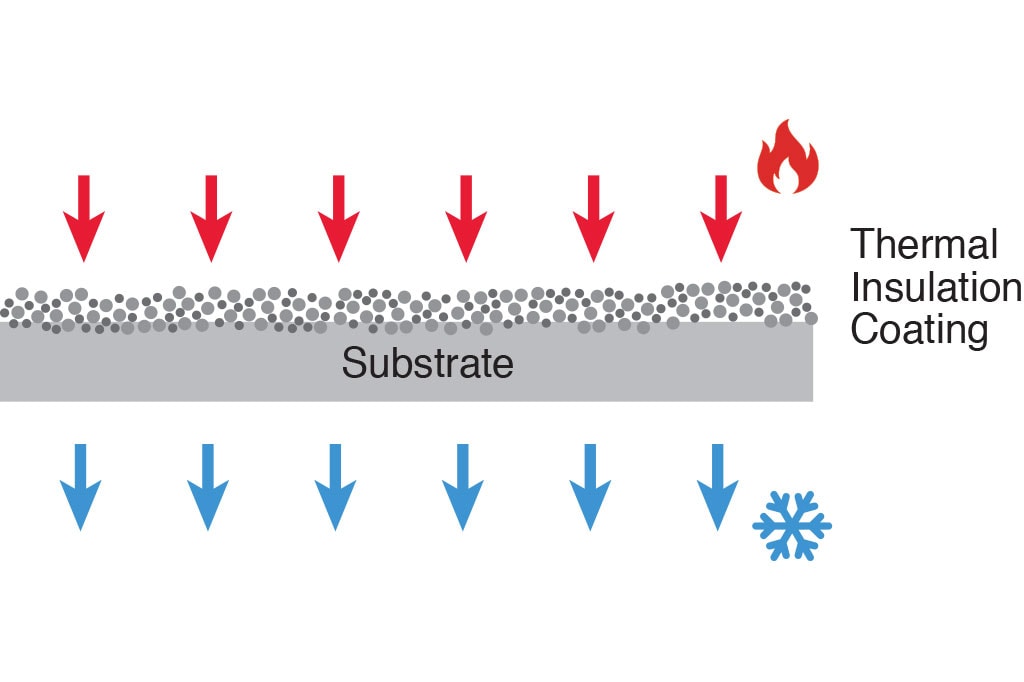
The health of patients and medical personnel depends on sterile, high-quality healthcare devices. Such devices must be capable of withstanding heavy use, corrosive substances and harsh cleaners, high temperatures, and more. To improve their safety, durability, and functionality, medical equipment manufacturers rely on solutions like thermal spray coatings.
If you’re looking for a coating for medical devices, Fisher Barton has over three decades of experience developing and engineering effective surface coatings for the healthcare sector. Our thermal spray coatings imbue the surfaces of medical devices like implants and electrosurgical instrumentation with critical resistance properties for enhanced performance. Designing coatings from metals, ceramics, carbides, and other advanced materials, our material engineers can design coating solutions to handle your unique requirements for optimal wear, corrosion, thermal, and electrical protection.
Benefits of Thermal Spray Coating for Medical Devices
Medical and biomedical instrumentation benefits from thermal spray coatings in several ways. The coatings’ primary advantages include:
- Offering good thermal resistance for applications involving high heat
- Resisting wear, friction, abrasion, and corrosion
- Safeguarding against fretting, cavitation, and poor adhesion
- Shielding from electromagnetic interference (EMI) and radio frequency interference (RFI)
- Making a surface electrically conductive or providing insulative properties, when necessary
Wear Resistance
Thermal spray coatings are comprised of hard metals, carbides, or oxide ceramics, delivering surface hardness of up to 1500 Vickers. Once deposited on the surface of a medical device, these coatings significantly improve their wear resistance and enhance a component’s longevity.
Antimicrobial Properties
We can use thermal spray coating alloys containing high copper content to provide devices with antimicrobial surfaces, which is highly beneficial for the medical sector. Applicable for both small and large surface areas, antimicrobial coatings are successful at killing various harmful pathogens to reduce the spread of contagious viral or bacterial infections via medical devices.
Types of Oxide Ceramics Coating Solutions We Offer
At Fisher Barton, we have multiple oxide ceramics coating types available to fulfill your various needs. Our team offers the following for medical devices:
- EMI/RFI shield coatings. As the name suggests, these medical equipment coatings offer shielding properties to protect components of varied substrates from EMI and RFI.
- Dielectric coatings. Certain oxide ceramic coatings are electrically insulative, providing dielectric properties for medical device surfaces.
Why We’re Different
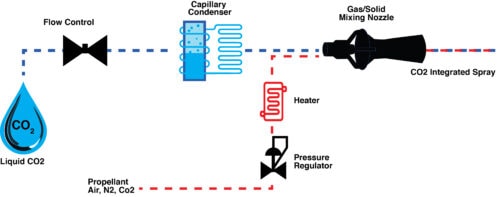
In addition to coating medical devices, Fisher Barton utilizes advanced technologies to provide medical device cleanings that ensure residue-free surfaces. An EPA- and FDA-approved cleaning method for medical devices, CO2 snow-jet cleaning is a non-aggressive, dry solution for eliminating residue from the surfaces of healthcare instruments, implants, and more for a sterile product.
Medical Device Coating Solutions by Fisher Barton
Selecting the right coating for medical devices is essential to maximizing their performance for improved patient care. Thermal spray coatings deliver a range of benefits, including increased component lifespan, corrosion resistance, and electrical insulation properties.
Specializing in high-wear, precision component manufacturing and metallurgy, Fisher Barton has delivered innovative and high-performance metal components to clients in various industries since 1906. Our metal, ceramic, and carbide coatings enhance medical devices’ quality and safety, and we use CO2 snow-jet cleaning technology to ensure a clean, sterile product without aggressive cleaners.
To speak with a specialist about our coating and cleaning solutions for medical devices and how Fisher Barton can support your business, contact us today.